Saliva has emerged as one of the most promising noninvasive sample types in modern molecular diagnostics. From oncology and infectious disease to liquid biopsy and precision medicine, saliva offers unparalleled convenience for patients and scalability for researchers. However, saliva is also a complex biological matrix that is rich in inhibitors, variable in viscosity, and challenging when it comes to isolating high-quality biomolecules. The solution doesn’t start with downstream assays; it starts with materials engineering.
Enabling Exosome and RNA Workflows Through Swab Collection Material Design
Extracellular vesicles (EVs), including exosomes, are nanoscale vesicles (approximately 30–150 nm) present in saliva and other biofluids. These vesicles carry RNA, DNA, and proteins reflective of biological state, making them valuable targets for molecular diagnostics.
Compared to whole-saliva analysis, EV-focused workflows can improve biomarker enrichment and reduce analytical interference. However, isolating EVs from saliva has traditionally required multiple cycles of refrigerated centrifugation and complex laboratory workflows, limiting scalability.
A comparison study conducted by Oasis Diagnostics® evaluated exosome recovery using the Porex saliva collection swabs in the Oasis Diagnostics® RNAPro•SalTM device versus a traditional centrifugation-based workflow.
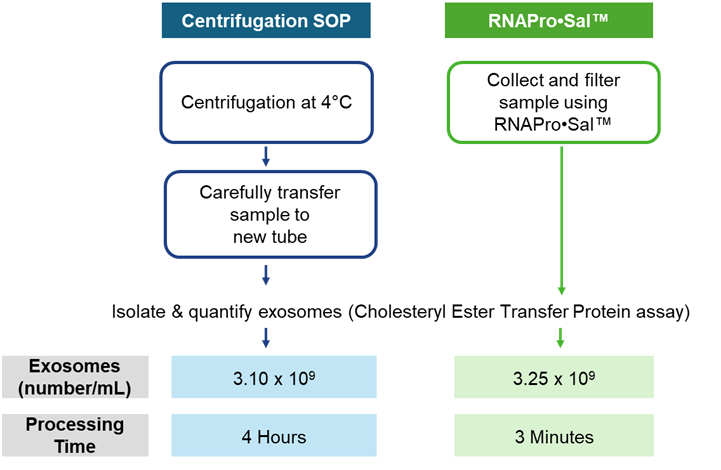
Figure 1. Comparison of Exosome recovery and Processing time for Centrifugation SOP vs RNAPro•SalTM
As shown in Figure 1, the RNAPro•SalTM produced similar exosome isolation to the standard centrifugation method, but with significantly less processing time required. Instead of the 4 hours and expertise required for centrifugation, the same results were achieved in just 3 minutes. These results show that comparable or improved exosome recovery can be achieved with the engineered saliva collection swab material while eliminating cold-chain requirements and reducing processing steps. This highlights how swab material design can simplify workflows while maintaining strong biomolecule recovery, and enable integration into research, clinical, and decentralized testing environments.
Built-In Inhibitor Retention for Cleaner Samples
Saliva-derived inhibitors such as mucins and excess proteins can interfere with EV quantification, nucleic acid isolation, and molecular amplification. In the same study described above, the material left behind after exosome extraction was pelleted for examination. RNAPro•SAL™ supernatants had visibly lower particulate matter vs. the centrifugation SOP, suggesting that the RNAPro•SAL™ device, with the engineered swab material, is removing these contaminants in the filter/device. These results show that an engineered saliva collection swab material can retain inhibitor materials within the porous structure, preventing their transfer into purified samples.
This material-driven mechanism delivers:
- Cleaner recovered samples
- Reduced assay interference
- Improved reproducibility across users and sites
By addressing inhibitors at the point of collection, variability is reduced before analytical testing begins.
Saliva Collection Swabs Designed for RNA Stability and Real World Use
Transport stability and nucleic acid preservation are essential for self-sampling diagnostic workflows. This is particularly relevant for RNA, which is fragile and prone to degradation. Standard operating procedures (SOPs) for isolating RNA from saliva require cold collection, refrigerated centrifugation, and careful pipetting, which take time and expertise to complete.
In a small study conducted by Oasis Diagnostics® with researchers at UCLA, the stability of RNA in samples collected using the RNAPro•SalTM system was evaluated to see if Porex engineered saliva collection swabs preserve extracellular RNA integrity in purified saliva samples stored at room temperature for extended periods. Saliva was collected from 6 patients and purified using the SOP and RNAPro•SalTM. RNase inhibitors were added to all purified samples, and then samples were stored for 0-14 days before RNA extraction and qPCR.
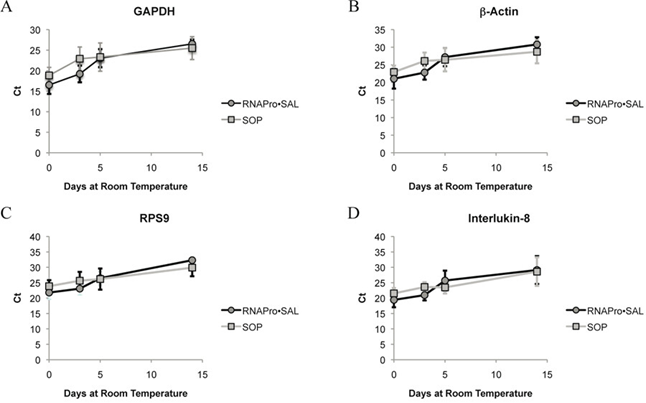
Figure 2. qPCR Results for the RNAPro•SalTM and SOP
The RNAPro•SalTM samples had comparable RNA yields and quality to the SOP. Figure 2 below shows consistent qPCR detection for key markers. These results show that RNA remains stable for up to 14 days when collected and processed using this engineered swab material. This demonstrates how material design can simplify workflows by integrating sample purification into the collection step itself and improve sample stability for downstream molecular analysis. This approach supports flexible transport and storage conditions, which is particularly important for decentralized and at-home sample collection models.
Saliva-based Diagnostics Performance Starts with the Swab Material
As molecular diagnostics continue to expand into self-collection and decentralized testing, controlling sample quality at the sample collection material level becomes increasingly important. Porex engineered swab materials provide a foundational solution for saliva-based diagnostics that enable exosome recovery without centrifugation, protects assays from inhibitors, and preserves sample RNA stability without the need for cold chain storage.
For saliva-based diagnostics and beyond, performance begins with collection swab material design. To see samples of Porex saliva swabs visit the sample selector here.
For more information, see our webinars on saliva diagnostics.

